Medicine Storage Audit Checklist UK: Care Home Compliance, Safety and Daily Checks Guide (2026)
May 6, 2026
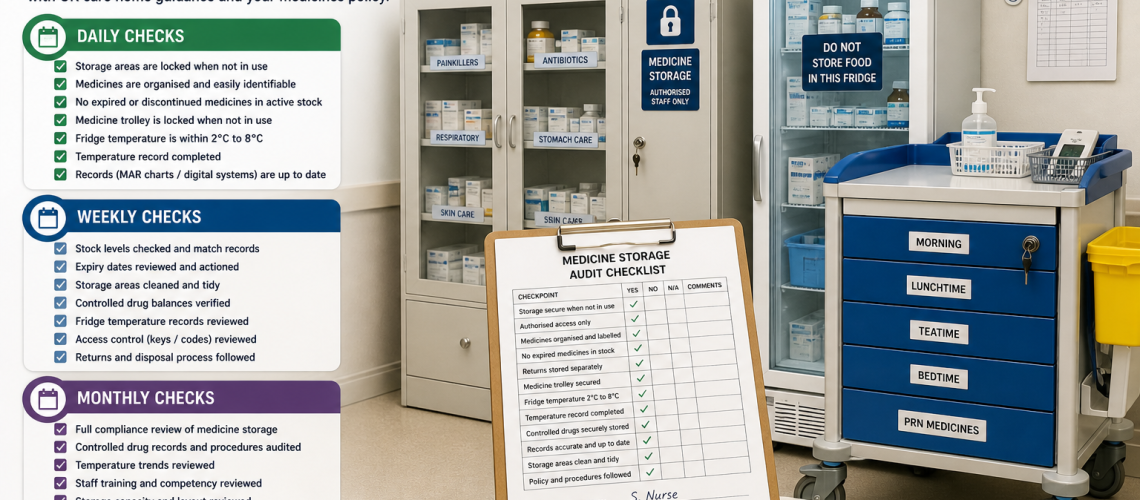
A medicine storage audit checklist helps UK care homes check that medicines are stored safely, securely and in line with daily procedures. It supports compliance, reduces errors and ensures medicines remain effective when administered.
This guide provides a complete care home medicine storage audit checklist. It covers cabinets, medicine rooms, trolleys, controlled drugs, fridge storage, temperature checks, access control, stock management and record keeping.
Start here: This page forms part of the Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, secure and manage medicine storage systems in UK workplaces and care settings.
Start here: This page forms part of the Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, secure and manage medicine storage systems in UK workplaces and care settings.
This audit checklist reviews existing medicine storage. For cabinet choice, secure access and the full storage framework, use the Medical Cabinets UK: Medicines Storage, Compliance and Security Guide.
Start here: This page forms part of the main Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, plan and manage secure medicine storage.
This checklist helps review medicine storage after a setup is in place. For planning, see medicine storage systems UK. For the main pillar, see the medical cabinets and medicine storage systems guide.
This audit checklist helps review medicine storage after a system is in place. For setup planning, see medicine storage systems UK. For care-home-specific storage, see care home medicine storage.
Use this checklist for daily, weekly and monthly audits. For full setup guidance, see the care home medicine storage guide, medicine room design guide and medical cabinets UK guide.
Quick answer: what should a medicine storage audit check?
A medicine storage audit should check security, access control, correct storage conditions, stock organisation, expiry dates, separation of medicines, fridge temperature, controlled drug storage, record accuracy and overall compliance with the care home’s medicines policy.
Daily medicine storage audit checklist
- Medicine storage areas are locked when not in use.
- Only authorised staff have access.
- Medicine cabinets are closed, secure and not overcrowded.
- Labels are clear and readable.
- No loose or unidentified medicines are present.
- Expired medicines are removed from active storage.
- Returned medicines are separated clearly from current stock.
- Medicine trolley is locked when not in active use.
- Trolley contents are organised and easy to check.
- Work surfaces are clean and clear.
- Medicine fridge temperature is recorded.
- Fridge contents are organised and not overloaded.
- Room temperature is within acceptable range where monitored.
- Records (MAR charts or digital systems) are complete and up to date.
Weekly audit checklist
- Full stock check completed against records.
- Expiry dates reviewed across all medicines.
- Medicine storage areas cleaned thoroughly.
- Cabinets checked for damage or faults.
- Trolley wheels, brakes and locks checked.
- Fridge temperature records reviewed for consistency.
- Controlled drug balances checked and verified.
- Returns and disposal processes followed correctly.
- Access control reviewed (keys, codes or permissions).
Monthly audit checklist
- Full compliance review against care home medicines policy.
- Audit of controlled drug procedures and records.
- Temperature monitoring trends reviewed.
- Staff adherence to storage procedures assessed.
- Storage capacity reviewed (overcrowding or underuse).
- Medicine room layout reviewed for risks or inefficiencies.
- Training needs identified for staff handling medicines.
- Audit findings recorded and actions tracked.
Medicine cabinet audit checks
- Cabinets are securely fixed where required.
- Locks are working and keys are controlled.
- Shelves are organised and labelled.
- No overcrowding or hidden stock.
- Separate storage for different residents or medicine types where needed.
- Clean and easy-to-maintain surfaces.
Medicine trolley audit checks
- Trolley is locked when not in use.
- Drawers or trays are clearly organised.
- Resident separation is clear.
- No mixing of returned and active medicines.
- Castors and brakes function correctly.
- Trolley is stored in a secure location.
Controlled drug storage audit
- Controlled drugs are stored separately.
- Access is restricted to authorised staff.
- Controlled drug register is accurate and complete.
- Stock balances match records.
- Storage location is secure and not exposed.
Medicine fridge audit
- Fridge temperature is recorded daily.
- Temperature is within required range (typically 2°C to 8°C where specified).
- Fridge is not overloaded.
- No food or unrelated items are stored.
- Fridge is clean and organised.
- Temperature issues are escalated and recorded.
Common audit failures in care homes
- Unlocked cabinets or trolleys.
- Expired medicines still in active storage.
- Poor separation between current and returned medicines.
- Missing or incomplete temperature records.
- Overcrowded cabinets or trolleys.
- Weak key or access control.
- Inaccurate or incomplete records.
- Fridge misuse or poor positioning.
Printable audit checklist (summary)
Use this quick checklist for daily audits:
- ✔ Storage locked
- ✔ Access controlled
- ✔ Medicines organised
- ✔ Expiry dates checked
- ✔ Fridge temperature logged
- ✔ Records updated
- ✔ Returns separated
- ✔ Trolley secured
If the audit finds storage, access or record-keeping issues, use the care home medicine storage risk assessment to identify the risk level and corrective action.
If your audit finds unsuitable cabinets, weak access control or unclear separation, use the care home medicine storage risk assessment and then return to the medical cabinets UK guide.
How this page fits
This page supports compliance and audit intent. It should link to:
- Care home medicine storage UK (rules and systems)
- Medicine room design (layout)
- Medical cabinets UK (products)
- Medicine trolleys UK (mobile storage)
If your audit finds storage, access or record-keeping issues, use the care home medicine storage risk assessment to identify risk level and corrective action.
Related medicine storage checks
- Care home medicine storage risk assessment
- Medical storage compliance UK
- Medical cabinets and medicine storage systems guide
- Medicine storage systems UK
Next step: Return to the Medical Cabinets UK guide to choose the correct storage setup.
Next step: Return to the Medical Cabinets UK guide to choose the correct storage setup.
FAQ: Medicine storage audits UK
How often should a care home audit medicine storage?
Most care homes complete daily checks, weekly stock reviews and monthly compliance audits. Frequency may vary based on policy and risk level.
What is checked during a medicine storage audit?
Audits check security, access control, storage conditions, organisation, expiry dates, records, fridge temperature and controlled drug storage.
Who is responsible for medicine storage audits?
Responsibility usually sits with trained staff such as senior carers, nurses or managers, depending on the care home’s structure and policy.
What happens if a medicine audit fails?
Failures should be recorded, investigated and corrected. Actions may include retraining staff, improving storage systems or updating procedures.
Do CQC inspections check medicine storage?
Yes. CQC inspections review how medicines are stored, handled and managed to ensure people receive safe care and treatment.
Discover more from Blog Total Locker Service
Subscribe to get the latest posts sent to your email.