Care Home Medicine Storage Risk Assessment Guide UK: Hazards, Controls and Safe Storage Planning (2026)
May 6, 2026
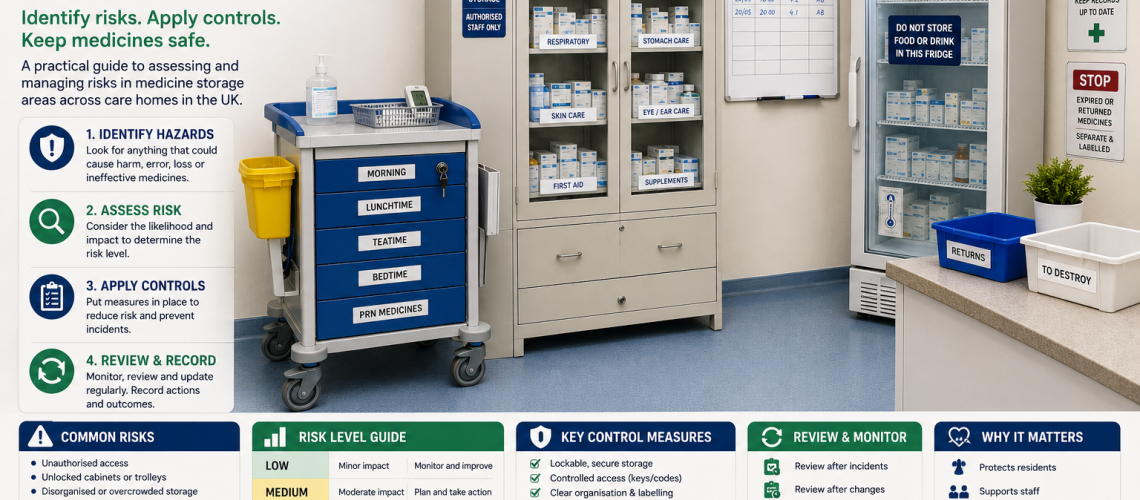
A care home medicine storage risk assessment identifies hazards, evaluates risks and sets controls to keep medicines secure, effective and safely managed. It supports compliance, protects residents and reduces avoidable errors.
This guide explains how to assess risks in care home medicine storage. It covers cabinets, medicine rooms, trolleys, controlled drugs, fridge storage, access control, temperature risks and common failures.
Start here: This page forms part of the Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, secure and manage medicine storage systems in UK workplaces and care settings.
Start here: This page forms part of the Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, secure and manage medicine storage systems in UK workplaces and care settings.
This risk assessment supports care home medicine storage and should be read alongside the main Medical Cabinets UK: Medicines Storage, Compliance and Security Guide.
Start here: This page forms part of the main Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, plan and manage secure medicine storage.
This risk assessment guide supports care home medicine storage and links back to the main medical cabinets and medicine storage systems guide.
This risk assessment guide supports care home medicine storage. For the wider storage framework, see the medical cabinets and medicine storage systems guide.
Use this page alongside the medicine storage audit checklist, care home medicine storage guide and medicine room design guide to build a complete compliance system.
Quick answer: what is a medicine storage risk assessment?
A medicine storage risk assessment identifies what could go wrong, how likely it is, how serious the outcome could be and what controls are needed to reduce risk. It should be documented, reviewed and linked to the care home’s medicines policy and audit process.
Step 1: Identify medicine storage hazards
Start by identifying anything that could lead to unsafe storage, incorrect administration or loss of medicine effectiveness.
- Unauthorised access to medicines.
- Unlocked cabinets or trolleys.
- Overcrowded or disorganised storage.
- Expired medicines in active stock.
- Mixing returned and current medicines.
- Poor temperature control.
- Fridge failure or incorrect use.
- Controlled drugs stored incorrectly.
- Incorrect labelling or unclear identification.
- Staff using inconsistent storage practices.
Step 2: Assess risk level
Each hazard should be assessed based on likelihood and impact. This helps prioritise which risks need urgent action.
| Risk level | Example | Action |
|---|---|---|
| Low | Minor organisation issue | Monitor and improve |
| Medium | Inconsistent labelling | Correct and review procedures |
| High | Unlocked medicine storage | Immediate action required |
| Critical | Controlled drug discrepancy | Urgent investigation and escalation |
If the assessment identifies weak access control, poor cabinet choice or unclear storage procedures, return to the medical cabinets UK guide before selecting replacement storage.
If the assessment identifies weak access control, unsuitable cabinets, poor separation or missing records, review medical storage compliance UK and the medicine storage audit checklist UK.
If the risk assessment identifies weak access control, poor separation, unsuitable cabinets or missing records, review the medical storage compliance UK guide and the medicine storage audit checklist UK.
Step 3: Apply control measures
Controls reduce the risk identified. These should be practical, consistent and built into daily routines.
- Lockable medicine cabinets and secure storage.
- Controlled access to keys, codes or systems.
- Clear storage separation (current, returns, controlled drugs).
- Temperature monitoring and recording.
- Medicine fridge use and maintenance.
- Regular stock checks and expiry reviews.
- Clear labelling and organisation systems.
- Staff training and supervision.
- Defined medicine room layout and workflow.
Step 4: Review storage areas
Risk assessment should cover every storage location, not just the main medicine room.
- Medicine rooms.
- Medicine cabinets in resident areas.
- Medicine trolleys used during rounds.
- Controlled drug storage locations.
- Medicine fridges.
- Temporary storage areas during delivery or returns.
Related risk and compliance guides
- Care home medicine storage
- Medicine storage audit checklist UK
- Medical storage compliance UK
- Medicine storage systems UK
Step 5: Document and act
Risk assessments must be recorded clearly. Actions should be assigned, completed and reviewed. A risk assessment that is not acted on has no value.
- Record identified risks.
- Assign responsibility for actions.
- Set review dates.
- Link to audit findings.
- Update procedures where needed.
Common medicine storage risks in care homes
- Storage areas left unlocked.
- Weak key or access control.
- Overfilled cabinets or trolleys.
- Temperature monitoring not completed.
- Fridge used incorrectly.
- Controlled drug records not matching stock.
- Staff not following procedures consistently.
- Poor separation of medicine types.
Risk assessment checklist
- ✔ Storage secured
- ✔ Access controlled
- ✔ Medicines organised
- ✔ Expiry dates managed
- ✔ Temperature controlled
- ✔ Records accurate
- ✔ Staff trained
- ✔ Procedures followed
Start here: This page forms part of the Medical Cabinets UK: Medicines Storage, Compliance and Security Guide, which explains how to choose, secure and manage medicine storage systems in UK workplaces and care settings.
Next step: Return to the Medical Cabinets UK guide to choose the correct storage setup.
Next step: Return to the Medical Cabinets UK guide to choose the correct storage setup.
FAQ: Care home medicine storage risk assessment
What is a medicine storage risk assessment in a care home?
It is a structured process to identify risks in how medicines are stored and to apply controls that reduce those risks.
Who should complete a medicine storage risk assessment?
Trained staff such as managers, nurses or senior carers usually complete or review risk assessments depending on the care home’s policy.
How often should medicine storage risks be reviewed?
Risk assessments should be reviewed regularly, especially after incidents, audit failures, changes in layout or changes in medicine procedures.
What are the biggest risks in medicine storage?
Common risks include unauthorised access, poor temperature control, expired medicines, incorrect storage and weak record keeping.
How does risk assessment link to audits?
Risk assessment identifies potential problems. Audits check whether controls are working. Both should work together as part of the same system.
Is a medicine storage risk assessment required for CQC compliance?
CQC expects care homes to manage risks effectively. A documented risk assessment supports safe care, governance and compliance.
Discover more from Blog Total Locker Service
Subscribe to get the latest posts sent to your email.